In this comprehensive article, we delve into the intricacies of Left Ventricular Systolic Dysfunction (LVSD), thoroughly analyzing its symptoms, causes, and severity. We explore the latest advancements in diagnostic tools and real-world case studies that demonstrate how AI-powered tools are enhancing the detection and management of LVSD. Join us as we highlight the critical aspects of this condition and the innovative technologies revolutionizing its diagnosis and treatment.
What is Left Ventricular Systolic Dysfunction (LVSD)?
Left ventricular systolic dysfunction (LVSD) is characterized by the heart’s inability to pump blood efficiently. The left ventricle, the heart’s main pumping chamber, fails to contract effectively, leading to reduced blood flow throughout the body. This condition can significantly impact overall health and quality of life. When symptoms occur, it is referred to as systolic heart failure or heart failure with reduced ejection fraction (HFrEF).
LVSD Symptoms
LVSD symptoms include shortness of breath, fatigue, and swelling in the legs and ankles. These symptoms result from the heart’s reduced pumping ability, leading to fluid retention and decreased oxygen delivery to tissues. Recognizing these signs early is essential for timely diagnosis and intervention.
What Causes LVSD?
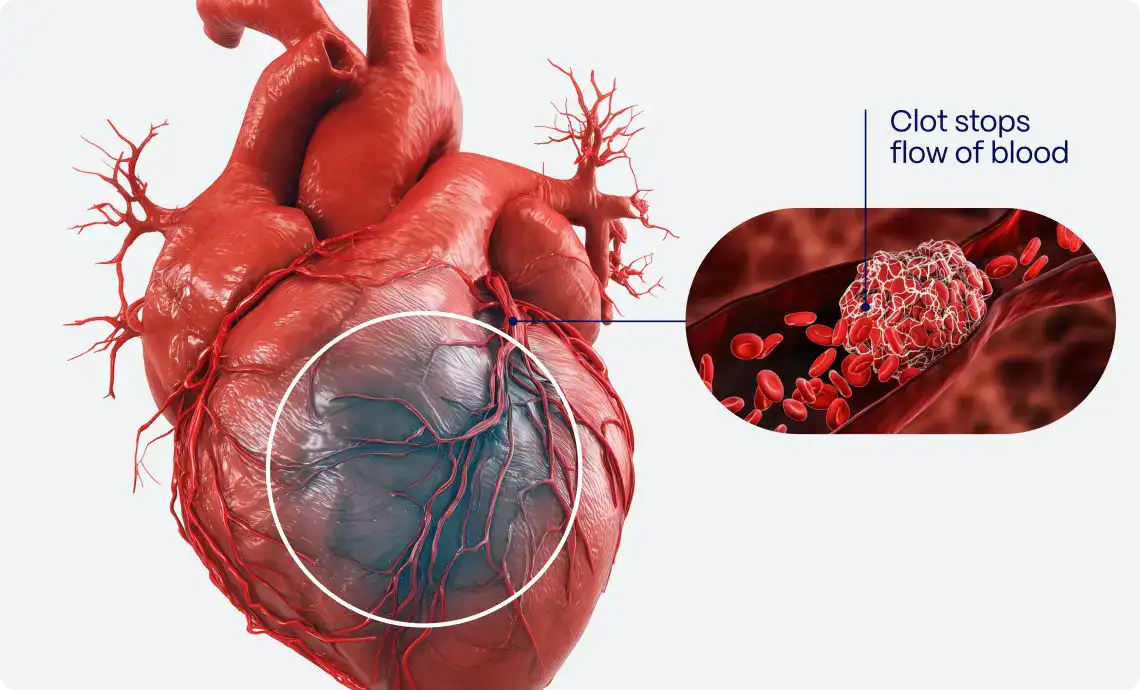
Several factors can lead to LVSD, including coronary artery disease, hypertension, and previous heart attacks. These conditions damage the heart muscle, reducing its ability to contract effectively. Other causes include cardiomyopathy, valvular heart disease, and chronic alcohol abuse. Understanding these underlying causes helps tailor treatment to address specific issues effectively.
How Serious is LVSD?
LVSD is a serious condition that can lead to significant health complications if left untreated. It increases the risk of heart failure, arrhythmias, and sudden cardiac death. Early diagnosis and management are essential to improve affected individuals’ outcomes and quality of life.
Types of Left Ventricular Systolic Dysfunction
Asymptomatic LVSD
Asymptomatic LVSD occurs when the left ventricle’s function is impaired, but the patient does not exhibit clear symptoms. This type is often detected incidentally during routine examinations or diagnostic tests for other conditions.
Moderate LVSD
Moderate LVSD is characterized by a noticeable decrease in the heart’s pumping ability, leading to symptoms such as shortness of breath and fatigue during moderate physical activity. It requires medical intervention to prevent progression.
Severe LVSD
Severe LVSD represents a significant impairment in the heart’s function, causing symptoms at rest or with minimal exertion. It often necessitates aggressive treatment strategies, including medication, lifestyle changes, and possibly surgical interventions.
How Do You Detect Left Ventricular Systolic Dysfunction?
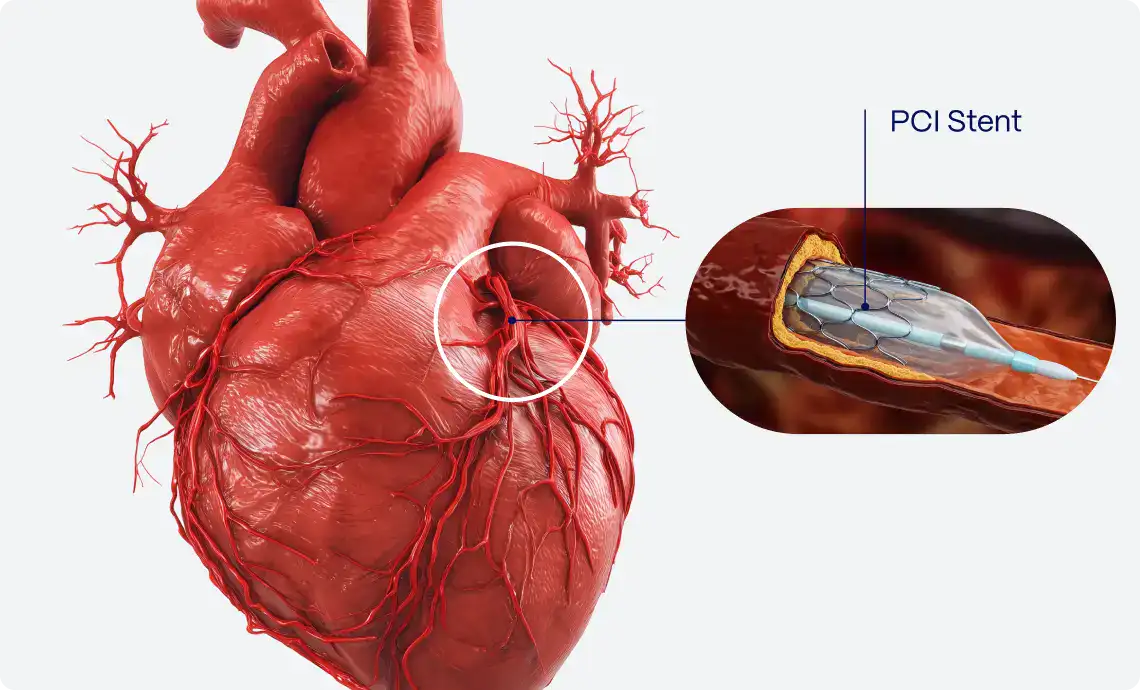
Detection of LVSD typically involves several diagnostic tools. An echocardiogram is the primary test, providing detailed images of the heart’s structure and function. One of the key measures obtained from an echocardiogram is the left ventricular ejection fraction (LVEF), which quantifies the percentage of blood the left ventricle pumps out with each contraction. Normal values for LVEF range from 55% to 70%. A reduced LVEF indicates compromised heart function, helping to diagnose LVSD. Electrocardiograms (ECG) are also used to assess the heart’s electrical activity and identify abnormalities indicative of LVSD.
AI ECG: Left Ventricular Systolic Dysfunction
Advancements in artificial intelligence (AI) have revolutionized the detection of LVSD. AI-powered ECG interpretation tools, like PMcardio, can quickly and accurately identify LVSD on a 12-lead electrocardiogram (ECG). These systems use machine learning algorithms to analyze patterns in the ECG, providing clinicians with valuable insights and improving diagnostic accuracy.
The following clinical cases demonstrate the practical application of the PMcardio AI Model in various scenarios of left ventricular systolic dysfunction. These examples highlight how AI-powered ECG analysis can enhance early detection, facilitate accurate diagnosis, and guide appropriate management strategies for patients with different degrees of LVSD.
ECG Case 1: Asymptomatic LVSD
An asymptomatic 69-year-old male underwent routine ECG screening at his GP. AI-ECG analysis detected reduced left ventricular function, prompting further echocardiographic evaluation. The follow-up echocardiogram confirmed the presence of early-stage left ventricular systolic dysfunction (LVSD), as evidenced by a left ventricular ejection fraction (LVEF) of 45%. These findings suggest the need for ongoing monitoring and potential intervention to manage and mitigate the progression of LVSD.
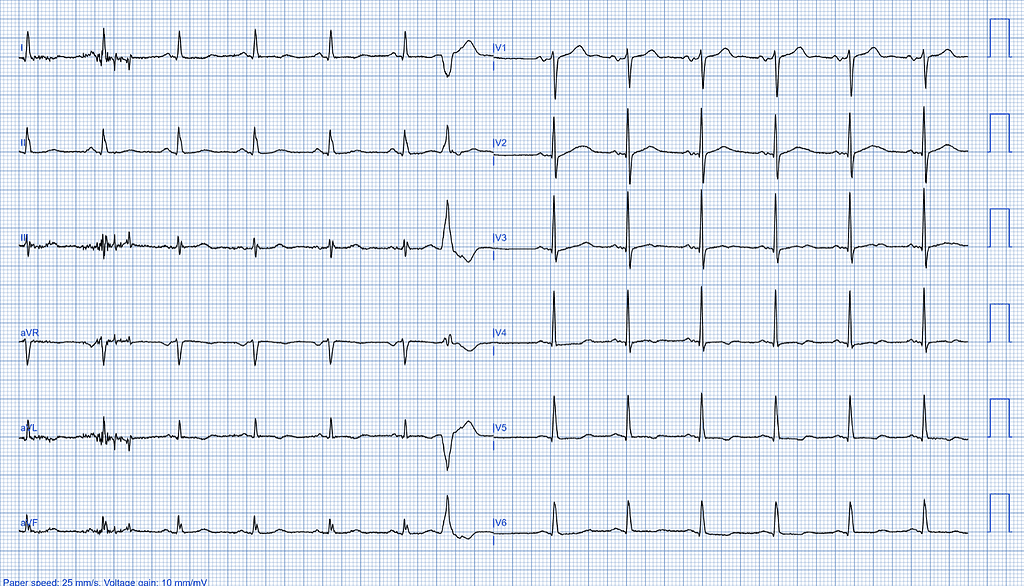
Initial ECG Findings
The initial ECG findings show a sinus rhythm, typically indicating the heart’s normal electrical activity. However, there is also the presence of a premature ventricular complex. Additionally, the ECG shows a nonspecific T wave abnormality, which may suggest an underlying issue requiring further investigation.
AI-ECG Analysis Results
The AI-ECG analysis results indicate reduced left ventricular function. The estimated left ventricular ejection fraction (LVEF) is between 41% and 49%, suggesting a moderate decrease in the heart’s pumping efficiency.
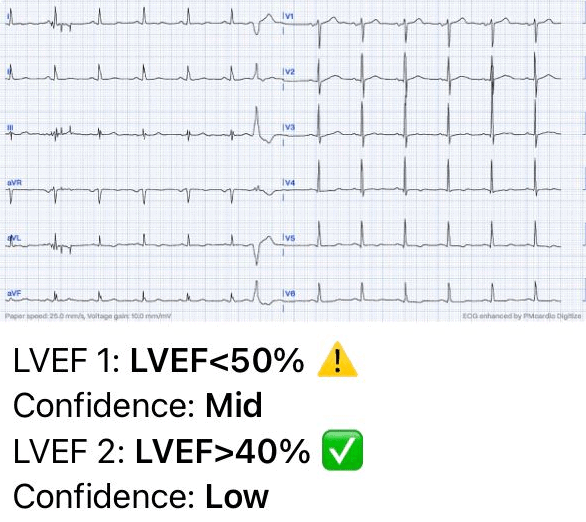
AI-ECG Analysis Result: Detected reduced left ventricular function. Estimated LVEF: 41-49% (Analyzed by PMcardio AI Model)
Echocardiographic Findings
The echocardiogram confirmed left ventricular systolic dysfunction with an LVEF of 46%, aligning with the AI-ECG findings. This consistency between the echocardiogram and AI-ECG results highlights AI capabilities in early LVEF diagnosis.
- Dimensions
- LVDd (2D): 53 mm
- LVDs (2D): 43 mm
- IVSd (2D): 9 mm
- LVPWd (2D): 8 mm
- LVEDV (2D): 162 ml
- LVESV (2D): 88 ml
- Left Ventricular Systolic Function
- LVEF: 46%
- Other Observations
- Mildly reduced LV systolic function
- Normal RV function
- Grade 1 diastolic dysfunction
- Mild left ventricular hypertrophy
- Mild mitral regurgitation
- Dilated atria
Recommendations:
A comprehensive management plan includes regular follow-up echocardiograms to monitor changes in left ventricular function. Emphasizing heart-healthy lifestyle choices, such as a nutritious diet and regular exercise, is crucial. Medical management may involve medications like ACE inhibitors to prevent adverse cardiac remodeling and manage blood pressure. Additionally, educating the patient about the worsening heart function symptoms and when to seek medical attention is essential for timely intervention.
Case Conclusion
The timely detection of asymptomatic LVSD through advanced AI-ECG analysis and subsequent echocardiographic confirmation underscores the importance of routine screening and early intervention in managing cardiovascular health. This proactive approach can help in preventing the progression of LVSD and improving patient outcomes.
ECG Case 2: Moderate LVSD
A 72-year-old woman presented to the Emergency Department with complaints of increasing shortness of breath. She states she has not experienced any chest pain. An ECG was promptly performed. The AI-ECG analysis indicated reduced left ventricular function, necessitating immediate further evaluation. The follow-up echocardiogram confirmed the presence of moderate left ventricular systolic dysfunction (LVSD), with a left ventricular ejection fraction (LVEF) of 35%.
At the Emergency Department, there is a need for swift and accurate diagnosis to manage the patient effectively. The AI-ECG analysis was instrumental in highlighting the reduced left ventricular function, which directed towards the necessity of echocardiographic confirmation.
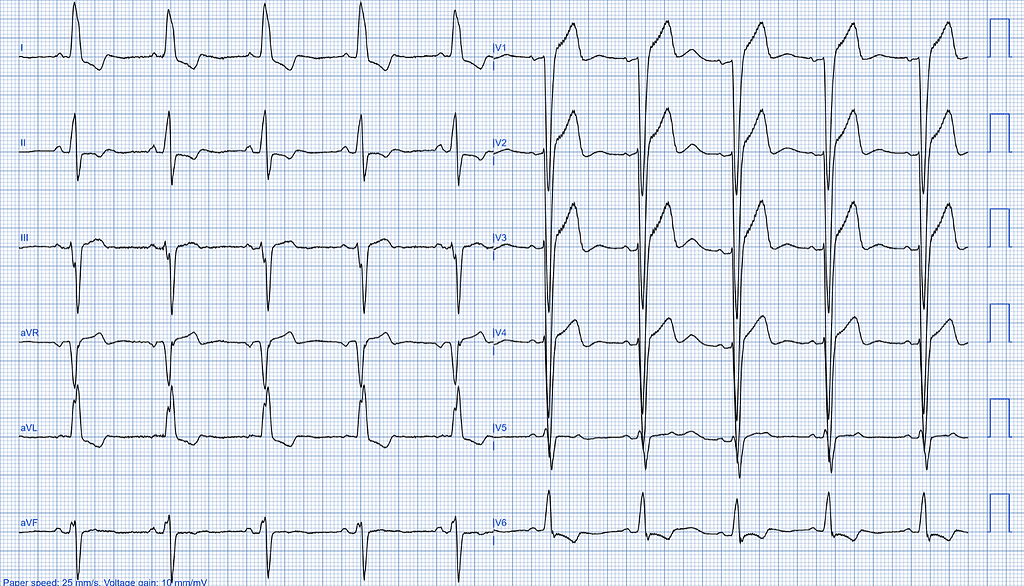
Initial ECG Findings
The ECG shows a sinus rhythm with left bundle branch block (LBBB), T wave abnormalities, and signs of left ventricular hypertrophy (LVH).
AI-ECG Analysis Results
The AI-ECG analysis results detected reduced left ventricular function, with an estimated left ventricular ejection fraction (LVEF) of less than 40%.
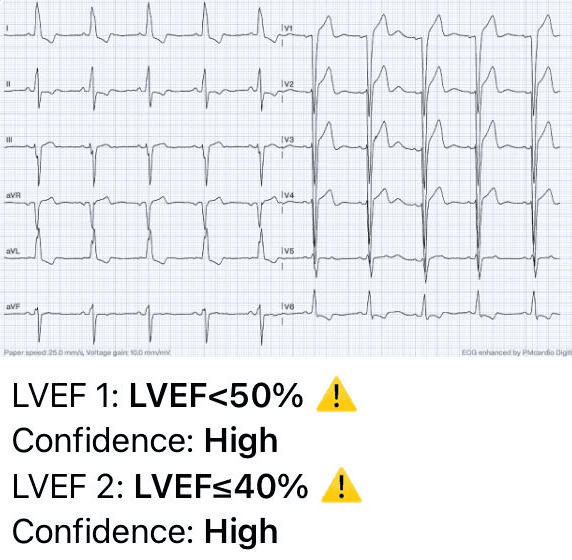
AI-ECG Analysis Results: Reduced left ventricular function. Estimated LVEF: <40%. (Analyzed by PMcardio AI Model)
Echocardiographic Findings
The echocardiogram confirmed moderate left ventricular systolic dysfunction, which aligns with the AI-ECG findings and further highlights AI capabilities in early LVEF diagnosis.
- Dimensions:
- LVDd (2D): 48 mm
- LVDs (2D): 40 mm
- IVSd (2D): 14 mm
- LVPWd (2D): 9 mm
- Left Ventricular Volumes:
- LVEDV (2D): 206 ml
- LVESV (2D): 130 ml
- Left Ventricular Systolic Function:
- LVEF: 37%
- Other Observations:
- Moderate left ventricular systolic dysfunction
- Grade 1 diastolic dysfunction
- Normal RV function
- Moderate mitral regurgitation
- Dilated atria
Clinical Insight
The use of advanced AI-ECG technology in the ED facilitated a quick and accurate assessment of the patient’s cardiac function. This case underscores the importance of integrating AI tools with traditional diagnostic methods to enhance the precision of initial evaluations and triage decisions. By accurately diagnosing LVSD, we could initiate timely and appropriate treatment.
Case Conclusion
In conclusion, the successful identification and confirmation of moderate LVSD in this patient highlight the vital role of AI-ECG in emergency medicine. It enables us to make informed decisions quickly, ensuring that patients receive the most effective care from the moment they arrive at the ED. This approach is essential for managing complex cardiac conditions and improving overall patient outcomes.
ECG Case 3: Normal Left Ventricular Function
A 53-year-old male presented to his General Practitioner (GP) with symptoms of fatigue and occasional shortness of breath. An ECG was performed and the patient was referred to a cardiologist for echocardiographic evaluation. The AI-ECG analysis reports normal LV systolic function on the ECG (LVEF>50%). The follow-up echocardiogram confirmed that the patient had a normal LVEF of 55%.
Initial ECG Findings
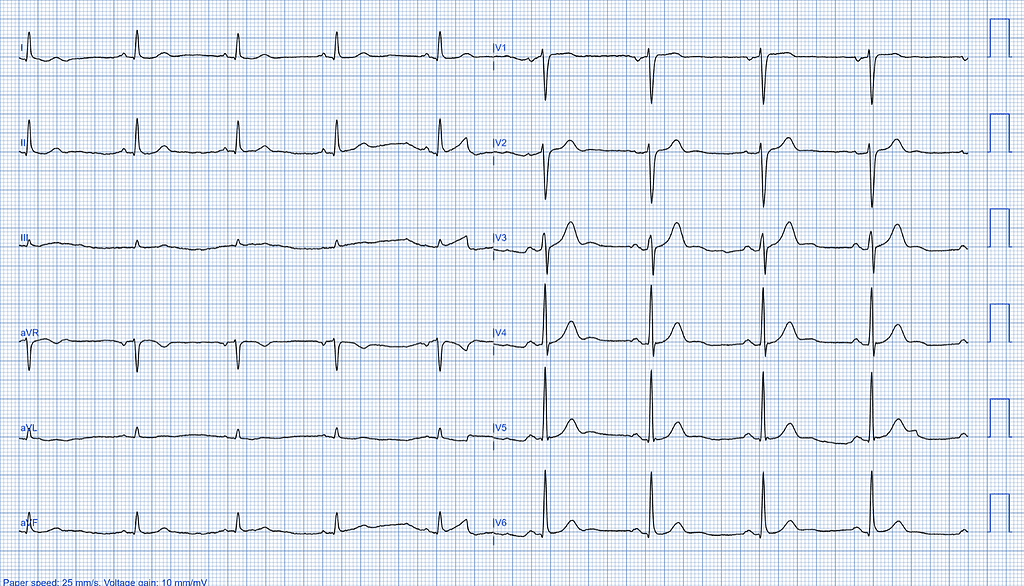
The ECG shows sinus bradycardia. Despite the reduced heart rate, all other parameters on the ECG appear normal, with no signs of structural or electrical abnormalities.
AI-ECG Analysis Results
The AI-ECG analysis reports normal left ventricular systolic function on the ECG, with an LVEF greater than 50%. However, it also suggests further evaluation due to presenting symptoms, indicating that while the systolic function appears normal, there may be other underlying issues requiring additional investigation.
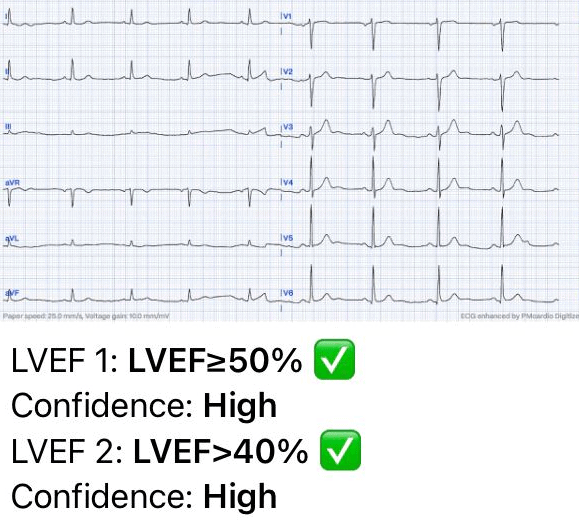
The AI-ECG analysis: Normal LV systolic function on the ECG (LVEF>50%) but also suggested further evaluation due to presenting symptoms. (Analyzed by PMcardio AI Model)
Echocardiographic Findings
The follow-up echocardiogram confirmed that the patient had a normal LVEF of 55%, aligning with the AI-ECG analysis reporting normal LV systolic function on the ECG (LVEF > 50%). This further highlights the AI’s capabilities in early LVEF diagnosis.
- Dimensions:
- LVDd (2D): 52 mm
- LVDs (2D): 31 mm
- IVSd (2D): 12 mm
- LVPWd (2D): 11 mm
- Left Ventricular Volumes:
- LVEDV (2D): 127 ml
- LVESV (2D): 57 ml
- Left Ventricular Systolic Function:
- LVEF: 55%
- Other Observations:
- Normal left ventricular systolic function
- No significant valvular abnormalities observed
- Normal RV function
- Normal atria
- Trace of mitral regurgitation
Case Conclusion
This case highlights the role of AI-ECG analysis even when LVEF is normal. The AI-ECG analysis can help with ruling out left ventricular systolic dysfunction as the cause of the symptoms and ensure appropriate management and follow-up for the patient.
ECG Case 4: Detection of cardiotoxicity in a Breast Cancer Patient during Chemotherapy Treatment
A 54-year-old woman was diagnosed with left-sided breast cancer in October 2015 and treated with chemotherapeutic agents, including Adriamycin, cyclophosphamide, and trastuzumab. She had a history of metastatic breast cancer treated with ADO–trastuzumab emtansine. An echocardiogram revealed a mildly reduced left ventricular ejection fraction (LVEF) of 48%, prompting evaluation for chemotherapy-induced cardiomyopathy. Her past medical history included hypertension, left bundle branch block (LBBB) on ECG, and hyperlipidemia.
CMR Analysis
A cardiac magnetic resonance (CMR) study using Displacement Encoding with Stimulated Echoes (DENSE) was performed. This advanced imaging technique enabled a detailed analysis of left ventricular (LV) contractile parameters, offering a more comprehensive assessment compared to traditional left ventricular ejection fraction (LVEF) measurements. The study revealed significant reductions in peak systolic longitudinal strain and torsion, confirming cardiac dysfunction.
AI-powered ECG Analysis
Our AI-ECG algorithm predicts reduced LVEF on the 12-lead ECG, already detecting the early signs of cardiotoxicity during treatment.
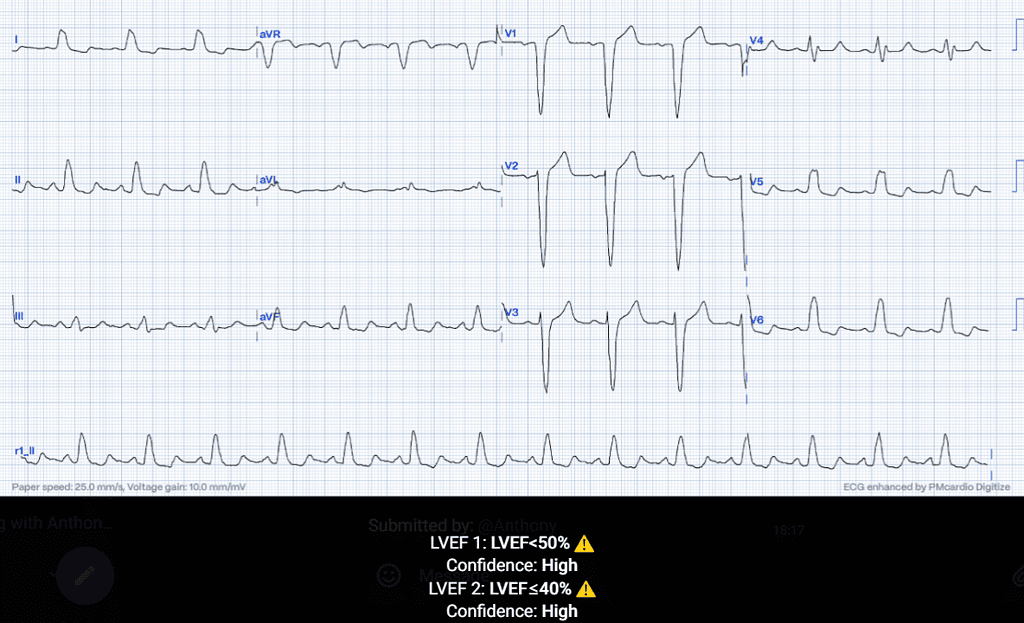
Case Conclusion
Cardiotoxicity was diagnosed based on the patient’s reduced LVEF, abnormal strain analysis, and the presence of multiple cardiac comorbidities. Cardiotoxicity is a likely diagnosis given the patient’s age, chemotherapy exposure, and the existence of cardiac comorbidities.
Furthermore, a follow-up echocardiogram showed an LVEF <25%. The findings highlighted the importance of continuous monitoring and advanced imaging techniques in detecting chemotherapy-induced cardiotoxicity early, improving long-term patient outcomes.
Perspective
This case underscores the value of advanced diagnostic tools like AI-ECG and CMR in identifying cardiotoxicity. Early detection allows for timely intervention, potentially mitigating the adverse effects of chemotherapeutic agents on cardiac function.
By integrating AI and advanced imaging into routine cardiac assessments, healthcare providers can enhance the precision of cardiotoxicity detection, ultimately leading to better patient care and outcomes.
Conclusion
Left ventricular systolic dysfunction is a serious condition with potentially severe health implications. Early detection and management are vital in improving patient outcomes. Advances in AI cardiology, such as AI-powered ECG interpretation, offer promising tools for enhancing diagnostic accuracy and facilitating timely interventions. Embracing these technological advancements can lead to better patient care and improved quality of life.